Exploring Immunogenic Peptides in CRISPR/Cas9: A Deep Dive for Researchers and Clinicians
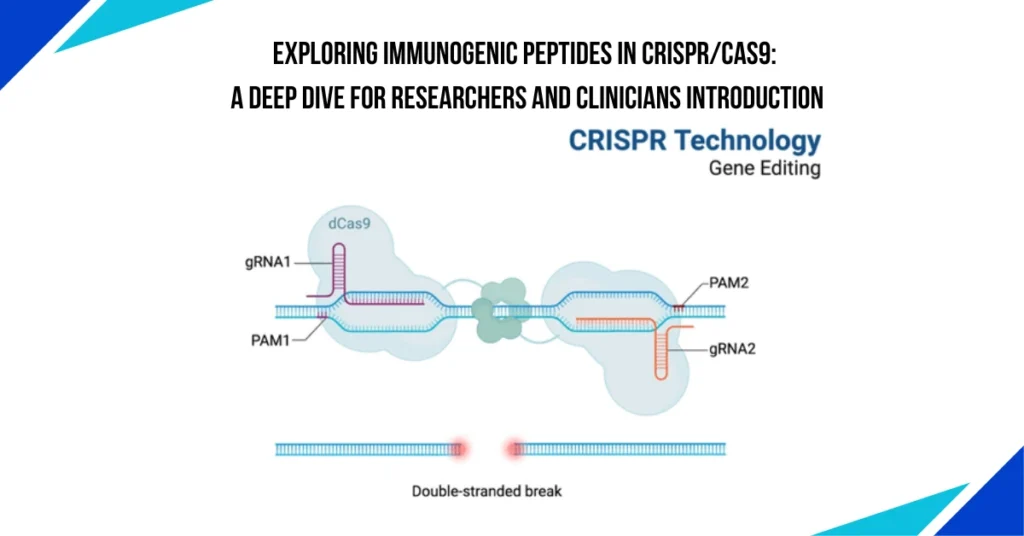
The CRISPR/Cas9 genome-editing technology has revolutionized the field of molecular biology, holding immense potential in gene therapy and medical treatments. However, despite its groundbreaking success, one of the critical challenges remains its safety, particularly related to the immune response it might trigger. Specifically, the immunogenic peptides generated during the CRISPR/Cas9 process have raised concerns regarding the technology’s clinical viability. Researchers have found that the Cas9 protein, especially when derived from bacterial species, can induce an immune reaction, limiting its use in humans.
We will explore the identification and characterization of immunogenic peptides associated with CRISPR/Cas9, highlighting their significance in clinical applications. We will discuss how these peptides might influence the immune response, how we can detect and mitigate them, and their potential impact on gene therapy protocols. By understanding the immunogenicity of CRISPR/Cas9, we can enhance the safety and efficacy of this technology for therapeutic use.
At Peptide Minds, we are dedicated to advancing the understanding and application of immunogenic peptides, particularly in CRISPR/Cas9 gene editing. Our expertise in peptide research and development ensures that we stay at the forefront of innovations aimed at optimizing gene therapy and minimizing immune responses. Visit our homepage for more insights and resources on how we’re pioneering peptide solutions in the world of molecular biology.
Immunogenicity in CRISPR/Cas9 Technology
What is Immunogenicity?
Immunogenicity refers to the ability of a substance, such as a protein or peptide, to induce an immune response. In the context of CRISPR/Cas9, this is a significant concern because the Cas9 protein, originally sourced from bacteria, can be recognized as foreign by the human immune system. This can lead to the production of antibodies and T-cell responses, causing unwanted immune reactions.
How Does CRISPR/Cas9 Trigger Immune Responses?
The Cas9 protein, used in genome editing, is often derived from Streptococcus pyogenes or Staphylococcus aureus. These bacterial proteins are seen as foreign invaders by the human immune system, which may respond by attacking the protein. This immune response can limit the efficacy of CRISPR/Cas9 therapies, especially when the therapy is administered multiple times. In addition, immune reactions could cause side effects like inflammation and tissue damage.
Overview of the Key Immunogenic Peptides
The immunogenic peptides of concern are small fragments of the Cas9 protein. These peptides are often recognized by immune cells, triggering an immune response. Research into identifying these peptides is ongoing, with various sequences of the Cas9 protein being tested for their ability to activate immune cells.
Role of Immunogenic Peptides in CRISPR/Cas9 Therapy
Clinical Implications of Immunogenic Peptides
Immunogenic peptides generated by the CRISPR/Cas9 process can pose significant challenges in clinical settings. In gene therapy, these peptides may lead to an immune response that affects the patient’s ability to tolerate repeated CRISPR treatments. This could be problematic, particularly in the case of chronic diseases where repeated gene editing is required.
Case Studies: Immune Responses in Animal Models
Researchers have observed immune responses to CRISPR/Cas9 in animal models, especially when the Cas9 protein from Streptococcus pyogenes was used. These responses were characterized by the development of antibodies against the Cas9 protein and inflammation at the site of delivery. Although these animal studies do not directly translate to human responses, they provide valuable insight into how the immune system might react in clinical trials.
Methods to Identify and Assess Immunogenic Peptides
Techniques to Uncover Immunogenic Peptides
To identify the immunogenic peptides generated by CRISPR/Cas9, several advanced techniques are employed. These include:
- Proteomics: A technique that involves analyzing proteins in a sample to identify the peptide sequences generated during CRISPR/Cas9 editing.
- Peptide Mapping: A method used to break down proteins into smaller fragments to study the sequence and determine which parts of the protein trigger immune responses.
- Bioinformatics: Computational tools that predict which sequences of the Cas9 protein might be immunogenic based on their structure and similarity to known immune-triggering sequences.
Role of Bioinformatics in Predicting Immunogenicity
Bioinformatics plays a crucial role in identifying potential immune-triggering sequences within the Cas9 protein. By using algorithms and databases that catalog known immunogenic peptides, researchers can predict which parts of the Cas9 protein are most likely to cause immune activation. This allows for early identification of problematic sequences and helps guide the development of safer CRISPR/Cas9 therapies.
Mitigating Immunogenic Reactions
Reducing Immune Responses to CRISPR/Cas9 Researchers have proposed several strategies to minimize the immunogenic response to CRISPR/Cas9:
Humanized or Modified Cas9 Proteins:
One approach is to create modified versions of the Cas9 protein that are less likely to provoke an immune response. These humanized versions are designed to be more similar to human proteins, making them less foreign to the immune system.
Alternative Delivery Methods:
Nanoparticles, lipid nanoparticles, and viral vectors can be used to deliver the CRISPR components in a way that minimizes immune activation. By encapsulating the Cas9 protein in a protective carrier, researchers can reduce its exposure to the immune system.
Immunosuppressive Treatments:
In some cases, temporary immunosuppression may be used to prevent the body from reacting to the Cas9 protein, especially when repeated treatments are needed.
Exploring Non-Immunogenic Cas9 Alternatives Other CRISPR-associated proteins, such as Cpf1 (now known as Cas12), have been explored as alternatives to Cas9. These proteins are derived from different bacterial species and may not provoke the same immune response. This opens up new possibilities for reducing immunogenicity in CRISPR-based gene therapies.
Future Directions in Immunogenicity Research for CRISPR/Cas9
Innovations in Immune Tolerance
Researchers are investigating methods to induce immune tolerance to CRISPR/Cas9 components. This includes developing protocols that gradually expose the immune system to the Cas9 protein, allowing the body to become accustomed to it without triggering a strong immune response.
Improving CRISPR/Cas9 Safety Profiles
Ongoing research into peptide engineering, immune-modulating agents, and novel delivery techniques will be essential for improving the safety profile of CRISPR/Cas9. These advancements will be crucial for advancing gene therapy into broader clinical use.
Integrating Immunogenicity Research into Clinical Practice
As we move closer to widespread clinical application of CRISPR/Cas9, understanding and mitigating the immunogenicity of the technology will be key. Continued research will help integrate these findings into clinical practice, ensuring CRISPR therapies are safe and effective for patients.
Conclusion
The immunogenic peptides associated with CRISPR/Cas9 technology present a significant challenge in its clinical adoption. By identifying and mitigating these peptides, we can enhance the safety and effectiveness of CRISPR-based therapies. Through innovative approaches like humanized proteins, alternative delivery methods, and immune-modulating strategies, the potential for CRISPR/Cas9 to revolutionize medicine remains strong. Ongoing research is critical to overcoming these immunogenic hurdles and unlocking the full potential of gene editing.
For those interested in exploring high-quality peptide products, visit the Peptide Minds shop for a range of solutions tailored to support cutting-edge research and clinical applications. Enhance your work with premium peptides that can drive innovation in your projects.
Frequently Ask Question
References
Jinek, M., et al. (2012). A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science, 337(6096), 816–821.
Liu, Q., et al. (2020). Immunogenicity of CRISPR/Cas9-based gene editing: A comprehensive review. Journal of Immunology Research, 2020, Article ID 9875891.
Wang, T., et al. (2018). Advances in CRISPR/Cas9 technology for human gene therapy. Journal of Clinical Investigation, 128(10), 4350–4360.
Zhang, J., et al. (2021). Immune response to CRISPR-Cas9 protein in clinical trials. Nature Reviews Drug Discovery, 20(2), 77–93.